At the intersection of technology, research and clinical care stands Verily’s Amy Abernethy. By Tanisha Carino and Jennifer Sukawaty.
As a Deputy Commissioner of the US Food and Drug Administration, Amy Abernethy won praise for modernizing the agency’s technology and data practices.
But just as speculation was rising that Abernethy would be chosen next to lead the agency, she departed to join Verily, the health tech venture of Alphabet, where she serves as President of Product Development and Chief Medical Officer. This newly created position puts product development under one leader versus multiple business unit presidents. In addition to product development, Abernethy leads clinical teams across Verily. Before joining the FDA in early 2019, Abernethy practiced oncology, taught at Duke medical school and helped lead a health tech startup. Twenty-seven triumphant months later, she joined Verily, formerly Google Life Sciences, a venture that boasts high ambitions and an all-star roster of talent.
Abernethy says her goals at Verily include improving the design of clinical studies, the process by which people participate in medical research and the productivity and efficiency of clinical trials. Abernethy spoke about her experiences and hopes at Verily with Brunswick Partner Tanisha Carino and Brunswick Director Jennifer Sukawaty.
Day to day at Verily, how are you influenced by your previous roles? You’ve worn so many hats—practicing clinician, professor of medicine, health tech executive, a leader of the FDA.
A close look at my career may suggest that I’ve done lots of different things. But zoom out, and you can see I’ve been focused on one problem, and I keep trying to find different ways of solving it.
When I was a melanoma doctor in a clinic, I was haunted by the idea that the person sitting in front of me was more likely to die than get access to treatments being discovered in laboratories on the other side of the wall.
Lowering that wall has been the focus of my career. We can’t just bring every prospective new treatment out of the lab and give it to patients, because we don’t know what’s effective, and we don’t know what’s safe.
But we have to build better, faster systems for improving the life of that person sitting in front of me, and technology can help us do that.
What’s at stake? What’s possible? Melanoma offers a glimpse. When I was in the melanoma clinic, the chance of a person with stage three melanoma dying in five years was 50%. These days, with BRAF inhibitors, and PD-1 drugs, and better ways of taking care of yourself and your immune system, we don’t even know the survival rates of melanoma. We just assume people are going to live a long time. We haven’t been able to redefine the end.
Your bio page on the Verily site states that you are determined to bridge the gap between clinical research and clinical care. What does that mean?
Could the research process be made more efficient? Could it be speedier? Could we get the best evidence into the healthcare delivery process more quickly so that, when we’re taking care of patients, we do the best of what we know?
Clinical research is conducted almost in a terrarium, under very controlled circumstances. We create an artificial world to get an answer that’s intended ultimately to be applicable to the real world. Meanwhile, in clinical care, we are trying to figure out how to match the best treatment available to an individual’s needs at that moment. Bridging the two can create not only economies of scale but also improved outcomes.
We can start to leverage data from clinical settings to inform research studies so that we reduce the data-collection burden and improve the efficiency of conducting research. This produces higher-quality information, which ultimately provides us better instructions about how to take care of patients.
The data sets that we need to inform research are inherently longitudinal. We live life across time. If we want to improve care delivery, we have to not only make clinical choices but update those choices as we monitor people across time.
When I was a melanoma doctor in a clinic, I was haunted by the idea that the person sitting in front of me was more likely to die than get access to treatments being discovered in laboratories on the other side of the wall.
Speaking of size, you sit inside an organization that has mastered scale. But how is that going to work for the individual who, for instance, goes to start chemo next Tuesday?
What’s exciting about the talent base, capabilities and horsepower behind Verily is that we can move toward something that is needed but that has been hard. That felt compelling to me.
But in healthcare you’ve got to start small. You’ve got to be humble enough to say, “You can’t always build for billions.” Sure, we have the resources and expertise to start making potentially significant improvements in the evidence generation process of clinical trials. The implications of that are big.
But the potential for small, important steps forward was clear during a recent visit to an eye clinic in Louisiana. We visited that clinic with a retinal camera, or fundus camera, which we developed with Google. The camera is very lightweight and easy-to-use and could support retinal imaging in retail and primary care settings. This camera does not require the patient’s eyes to be dilated, which for the patient adds significant time, cost and the inconvenience of limited vision for several hours. So, the camera would both save the optometrist and ophthalmology clinic time conducting retinal screenings and get adequate images to support diabetic retinopathy screening at scale by being adopted in a broader range of settings.
Google initially did ideation and user research around retinal cameras—how might we radically improve the access to retinal imaging and improve the patient and clinician experience? They brought this research to Verily, which we subsequently developed into what is now called the Verily Retinal Camera.
When this camera was brought to us to continue to develop and get ready to commercialize, it suddenly went from this idea of being able to develop an easy-to-use camera to asking the question, “Does this have commercial viability? Is this something that we can figure out what the reimbursement strategy around it would look like?” If you want things to work in healthcare, they have to be successful businesses, too.
So, it goes from this fairly research-based project at Google to something that, now, is quite tactical for Verily. When our team started to work on this camera, we figured out how to get a reimbursement code for it, how to make sure that it could be used in primary care practices. Then, we started it out at only a couple of clinics in the very beginning. Not only because we had a limited supply of cameras, but because we had to figure out how to best deploythem and teach the nurses.
Could we get the best evidence into the healthcare delivery process more quickly so that, when we’re taking care of patients, we do the best of what we know?
That all happened at the end of 2022. The camera worked. But, I said, “Yes, we want to continue to develop the camera. But let’s ask a bigger question: ‘How does the availability of pictures of the back of the eye now open up new capabilities as it relates to bridging research and care?’”
As you may have seen in the news recently, images from the back of the eye can identify a person at risk of several conditions—Alzheimer’s disease, neurologic disease, cardiovascular disease.
Now, we can start to imagine doing diabetic retinopathy screening and asking patients for permission to sign up for a registry, where we can follow them longitudinally and connect the dots between what we know about the picture from the back of the eye and how their diabetes is faring, or open up our understanding even beyond, to their risk of cardiovascular disease.
Then, how do we bring the AI teams into the story? If we’ve got people signed up to participate in longitudinal registries using our camera, willing to dedicate and donate their information, how can we put that to work for heart disease? And then, how do we connect that up to other products that we have in the suite?
There’s another technology that started at Google that we’re starting to use at Verily. The first part is something we developed here at Verily for research purposes. Our virtual stainer technology takes an unstained pathology slide, which is transparent, and turns them into digital images with H&E stain (the pink and purple ones that pathologists usually look at underneath the microscope).
This produces clinical findings just as vivid as if we’d stained the patient’s tissue with many different stains. It’s completely wild—this idea that you can literally peer inside the tissue and understand what’s going on without adding anything that disrupts the tissue, or changes the coloration, or anything else. We see virtual stainer as fundamentally changing pathology just like digital cameras removed the need for developing film.
Again using technology developed at Google, we now use AI to make new predictions that we couldn’t before—new ways of measuring whose disease might progress or not. Those two things were developed at Google as early stage ideas. Now, they come into Verily so we can work to bring them to market.
What advantage does being part of Verily offer you in terms of breaking down the walls between clinical research and practice? Let’s say, compared with your previous experience in academic or government medicine?
Sitting inside an academic health system, I had defined a problem I felt passionate about solving, but I topped out on two things. I topped out on access to talent, especially engineering, and I topped out on access to capital.
To do anything at scale, we needed those two things. The reason I went to Flatiron [the healthcare startup where she served as an executive before joining the FDA] was that those were the two things that I could see suddenly getting unlocked in totally different ways.
Now, talent, scale and capital are what make Verily so compelling.
Importantly, this isn’t just one kind of talent. It’s best-of-the-best software engineers, molecular scientists, data scientists, AI engineers, biostatisticians, clinical trial experts and medical clinicians. The difference between now and 10 years ago is that there’s a widespread understanding that these different pieces must come together.
You frankly just can’t have an engineering org-anization solve these kinds of big problems, any more than any one clinical organization could do it.
That’s something I learned at Flatiron: how to bring together these disparate specialists and experts, get them talking to each other and respecting each other.
Second thing I would say is you can’t just “technify” everything. Software isn’t enough. You need a services component. You need people in the system. At Verily, we have a spectacular team doing participant recruitment for clinical trials. And we have the best in digital marketing. Think about what we can do as a company with Google heritage.
But many of our patients who are being recruited to participate in clinical trials still need a warm phone call. That phone call and the warm introduction to a study coordinator at a site is what builds trust and answers the questions that real people have.
That’s hard to do with chatbots alone.
Almost two years into this role at Verily, is there anything you would do differently if you were starting over?
When I first arrived at Verily I was responsible for the clinical research business. I started with the mindset of focusing on my business line, the mindset of staying in my lane. Get my stuff done.
What I wish I’d done was a long walkabout to learn everything in all the different zones here at Alphabet and Verily. For example, I may have learned months, even years, earlier about the retinal camera. What I learned is to be curious across your organization and outside it.
By joining Alphabet during the pandemic, I missed out for some period of time on the value of meeting new people and learning new information from across the company. It’s common at tech companies to move so fast and focus so tightly that you also lose access to all of the great ideas that are happening outside your organization—you can also lose competitive intel.
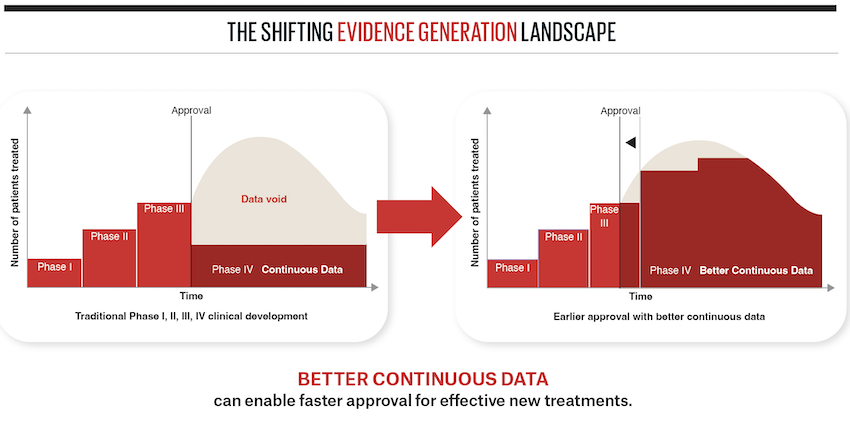
We should not have to wait forever to make the decision about whether a treatment is reasonably safe or effective.
The randomized controlled trial has been the gold standard for a long time, since long before the creation of the internet. Should it still be the gold standard?
The randomized controlled trial is a critical tool in our toolbox that does the important job of helping us understand if an intervention is more effective and safer than whatever it was that it was being compared to in the circumstances where that kind of comparison is required.
There are a number of different flavors of the randomized controlled trial. And, we have the opportunity of innovating on the future of randomized controlled trials.
You probably saw the RECOVERY trial, which was a UK-based platform trial for studying different interventions to treat COVID at the same time. They basically had one common control arm and then just kept studying a whole bunch of new treatments against that common control. Why randomize everybody one to one? Why not make it eight to one?
That’s one way to handle it. Another way to handle it is to do what needs to be randomized but rather than collect every data point anew, fill in the data set with data that has already been collected inother places. This way you preserve randomization and reduce the burden of conducting a trial for the patients and clinical site who are participating in the research. And that’s certainly one of the things I focus a lot on at Verily.
It’s important that we’re not lowering the evidence standard. We should be making sure that things work with enough confidence and credibility that we believe that it’s safe and effective enough to get in the hands of the public. Importantly, like we saw in COVID and in cancer care, these might be shorter or smaller clinical trials that get treatments into the clinic sooner.
We should not have to wait forever to make thedecision about whether a treatment is reasonably safe or effective. Instead, we can create systems that allow us to make a reasonably informed decision, and then continue to monitor things across time, and update the decisions that we’ve made by continuously cross-checking.
A good example here that’s playing out right now is cell and gene therapies, where there are life saving treatments. Many times, the effect size is huge, so you don’t even need a randomized trial to know if it works.
Why would we not want to get it into the hands of people with sickle cell and other diseases who need the treatment? Let’s figure out a path towards making that happen.
But also, let’s monitor that treatment for long periods of time to make sure that these viral vectors don’t go astray, or that the treatments continue to be safe enough or effective enough across time.
If we can come up with better systems to cross-check the decision that we make, then our anxiety about the perfect randomized controlled trial goes down, because we are confident that we will crosscheck the decision at later points in time.
More from this issue

People Puzzle
Most read from this issue

Critical Moment: Paris Olympics, 1924






